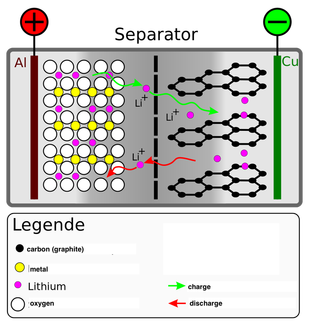
 modified after wikipedia.de modified after wikipedia.de Lithium-ion batteries are everywhere: in your smartphone, in your camera, … These types of rechargeable batteries are great for any portable electronics as it has a small memory effect and low self-discharge. However, that doesn’t mean that it can’t be improved. In 2007, Fu et al. tested three-dimensionally ordered mesoporous titania microparticles as “new” anode material. Why titania? I don’t know. Fact is, “conventional Li-storage materials suffer serious capacity loss when they are charged/discharged at a high rate. Polarization caused by slow diffusion of lithium ions and electrons in active materials is one challenging problem.“ As far as I understand it (and please my dear reader correct me if I am wrong) that means, that the lithium ions need a certain time to travel between between the electrodes. Therefore, if you are charging and discharging faster than the lithium ions need to travel from the one electrode to the other, this results in a capacity loss. Normally the anode in a lithium ion battery is made of graphitic layers. Between the stacked layers the lithium ions are normally stored in the charging process. While discharging, the lithium ions travel to the cathode. So summing up, the traveling time between the anode and the cathode (and the other way around) is the reason for polarization. However, “previous studies showed that electrode materials with three-dimensionally (3D) ordered mesoporous structure could decrease the polarization and enhance the performance at high rates”. That means that if the electrode material has a 3d structure with “pores” instead of the graphitic layers, the lithium ions can travel faster and so the polarization is decreased. Therefore, Fu et al. created a 3D ordered mesoporous structure out of titania and used it as anode. Indeed, the 3d structure was “beneficial to the diffusion of lithium ions” which reduced the capacity loss. So 3d is better than 2d… at least regarding the structure of the anode material in lithium ion batteries… (for movies I still prefer 2d… but this is maybe a question of taste). "Preparation and characterization of three-dimensionally ordered mesoporous titania microparticles as anode material for lithium ion battery."
Fu et al. Electrochemistry Communications 9.8 (2007): 2140-2144.
0 Kommentare
 The best “papers of the day” are papers which deal with problems I never thought about although they are quite obvious. This is the case for the paper of Amr M. Abd-Elhady et al. (2014) which deals with the problem of backflow current-overvoltages in wind turbines. I never thought about it, but wind turbines have a high risk of being hit by a lightning due to their size and location. Of course, wind turbines have a grounding system. However, that grounding system depends on the soil resistivity (= how much the soil resists the flow of electricity) which depends on moisture and salt content and temperature. High soil resistivity makes it “difficult to attain low grounding impedance for the utilized electrodes. These increase the ground potential rise (GPR) and make surge arresters operate in reverse direction.” The resulting backflow surge current can damage the equipment (cable, transformers, surge arresters . . .) and therefore needs to be prevented. Using a modelling approach, Amr M. Abd-Elhady et al. analysed which parameters influence the damage caused by lightning and tested an alternative grounding system. For example: The lightning damage is worse when the lightning hits the wind turbine at the negative peak of voltage waveform. Moreover, they compared the damage in “in service” and “out of service” wind turbines and the influence of the number of connected wind turbines in a wind farm. Interestingly, the damage decreases with increasing the number of wind turbines for the non-thunderstruck turbines case and increases with increasing the number of wind turbines for the thunderstruck turbines. So in larger wind farms, the wind turbine which is hit by lightning receives more damage but the surrounded wind turbines are less damaged compared to a small wind farm. High-frequency modeling of Zafarana wind farm and reduction of backflow current-overvoltages
Amr M. Abd-Elhady, Nehmdoh A. Sabiha and Mohamed A. Izzularab International Transactions on Electrical Energy Systems 24.4 (2014): 457-476. We do it every day… we like it when it is the coffee in the morning and don’t like it when it is a sweating person in the full subway. Our olfactory system detects all the pleasant and unpleasant odours of our environment for 24h every day of our life. However, what seems so simple in our daily life is quite hard to rebuild with electronic-chemical devices, so called electronic noses. In order to smell something, different kinds of odourant molecules needs to be detected and their relative amounts decide about the flavour they are creating. Julian W. Gardner and Philip N. Bartlett published 1994 a review about the history electronic noses. The first electronic noses were already build in the early 60’s but it needed further 20 years in order to improve an intelligent design for odour detection and the first conference for electronic noses was held in 1990. In the human nose, G-binding proteins act as chemosensory receptors in the olfactory cells. The binding of the odour molecules with the receptors triggers a second-messenger signal cascade which ends up in the creation of action potential which are transferred by nerve cells to the brain. In the electronic nose, inorganic semiconducting materials such as oxides and catalytic metals have been used as sensors for odour molecules. For example, the electronic nose of Wilkens and Hatman (1964) was based on redox reactions of the odourants at an electrode. The sensors are enough for odour detection, but for odour classification, the electric signal created by the receptors needs to be analysed by the usage of pattern recognition (PARC) engine or supervised learning artificial networking technique. The main problems in building electronic noses are the conflicts between sensitivity, selectivity and life duration. The sensitivity of the sensors depends on environmental properties such as temperature and humidity. Moreover, it is difficult to distinguish between the different odour molecules and reactive species decrease the life time of the sensors. However, the receptor cells in the human nose also have a low sensitivity, low specificity and short life duration (22 days). It is the subsequent neuronal processing which increases the sensitivity (by three orders magnitude) and offers us the probability to distingue between several thousand odours. So the solution for the electronic nose problems is not located in the odour sensors but the computation network behind it. A brief history of electronic noses
Julian W. Gardner and Philip N. Bartlett Sensors and Actuators B: Chemical 18.1 (1994): 210-211. |
IdeaI love to increase my general science knowledge by reading papers from different fields of science. Here I share some of them. Archiv
März 2018
Kategorien
Alle
|

 RSS-Feed
RSS-Feed
